
Lose Belly Fat Through Metabolism Reset
Abdominal fat accumulation reflects metabolic dysfunction, not cosmetic failure. Lose Belly Fat Through Metabolism Reset requires correcting hormonal signaling, mitochondrial energy use, and behavioral inputs that govern fat storage. Caloric math alone fails because visceral adipose tissue responds primarily to insulin exposure, sleep integrity, muscle demand, and chronic stress chemistry rather than short term dietary restriction.
The Biology of Visceral Fat Storage
Belly fat is predominantly visceral fat, stored deep around organs and strongly associated with cardiometabolic disease. Unlike subcutaneous fat, visceral fat is metabolically active, releasing inflammatory cytokines that interfere with insulin signaling and lipid regulation. Research summarized by the National Heart Lung and Blood Institute shows visceral adiposity correlates with elevated triglycerides, impaired glucose tolerance, and increased cardiovascular burden.
Adipocytes in the abdominal region exhibit higher concentrations of beta adrenergic receptors and glucocorticoid receptors. This receptor density makes them highly responsive to cortisol and chronically elevated insulin. When insulin remains elevated throughout the day, hormone sensitive lipase remains inhibited, preventing fat mobilization regardless of caloric intake.
Mitochondrial downregulation compounds the issue. Sedentary behavior reduces mitochondrial density in skeletal muscle, lowering fatty acid oxidation capacity. Data reviewed by the National Center for Biotechnology Information demonstrates that reduced oxidative capacity shifts the body toward glucose dependency, encouraging fat storage during even moderate carbohydrate intake.
Metabolic reset therefore begins with restoring metabolic flexibility, defined as the ability to alternate efficiently between glucose and fatty acid utilization.
Lose Belly Fat Through Metabolism Reset

Metabolic reset is a physiological recalibration. It is not detoxification, cleansing, or extreme dieting. It is the normalization of four regulatory systems:
- Insulin rhythm
- Muscle driven glucose disposal
- Circadian hormone timing
- Nervous system stress load
When these systems synchronize, stored fat becomes accessible fuel.
Chronically elevated insulin is the primary gatekeeper. Even modest hyperinsulinemia blocks lipolysis. Evidence published in Diabetes Care demonstrates that improving insulin sensitivity alone increases fat oxidation independent of calorie reduction.
Muscle tissue acts as the largest glucose sink. Resistance training increases GLUT4 transporter expression, allowing glucose clearance without excessive insulin release. This mechanism directly reduces abdominal fat deposition.
Circadian alignment regulates leptin and ghrelin secretion. Disrupted sleep shifts appetite signaling toward energy storage. Findings summarized by the Sleep Foundation show sleep restriction decreases insulin sensitivity within days.
Stress chemistry alters fat distribution. Cortisol mobilizes energy acutely but promotes central fat storage when persistently elevated. Chronic psychological load therefore manifests physically as abdominal adiposity.
Resetting metabolism requires behavioral inputs that reverse these signals consistently enough to retrain endocrine response.
Nutritional Patterns That Reduce Insulin Exposure
Frequent eating maintains insulin elevation. Reducing eating frequency without severe calorie restriction allows insulin to fall long enough for lipolysis to occur. This is metabolic spacing, not starvation.
Protein prioritization improves satiety signaling and thermogenesis. Dietary protein has a higher thermic effect than carbohydrate or fat, increasing postprandial energy expenditure. Analysis from the Harvard T H Chan School of Public Health highlights protein’s role in weight regulation and lean mass preservation.
Unprocessed foods improve hepatic insulin sensitivity because they reduce rapid glucose excursions. Ultra processed foods combine refined starch, industrial fats, and sodium in ways that override satiety mechanisms. Observational data discussed by the World Health Organization links processed dietary patterns to increased obesity prevalence globally.
Fiber intake alters gut derived metabolic signaling. Soluble fiber slows glucose absorption and feeds short chain fatty acid production in the colon, which improves insulin sensitivity. Reviews in Nutrients describe how microbiome fermentation products regulate energy homeostasis.
Hydration status also influences fat metabolism. Even mild dehydration raises cortisol and reduces cellular efficiency. Adequate water intake supports lipolysis through improved circulation and enzymatic activity.
The objective is hormonal quieting, not caloric punishment.
Resistance Training as a Metabolic Switch
Aerobic exercise alone does not sufficiently counteract visceral fat because it does not create lasting metabolic demand. Resistance training transforms skeletal muscle into an endocrine organ that actively regulates metabolism.
Contracting muscle releases myokines, signaling molecules that enhance fat oxidation and reduce systemic inflammation. Research reported in Frontiers in Physiology identifies interleukin six released during resistance exercise as a mediator of improved glucose regulation.
Muscle mass increases resting metabolic rate because lean tissue requires constant energy turnover. Each additional kilogram of muscle raises basal energy expenditure, shifting the body toward continuous fuel use rather than storage.
Strength training also improves insulin independent glucose uptake. This allows carbohydrates to replenish glycogen instead of converting to fat. Guidelines from the Centers for Disease Control and Prevention emphasize muscle strengthening activity for metabolic health, not solely cardiovascular conditioning.
Progressive overload is required. Without increasing mechanical demand, adaptation halts and metabolic improvement plateaus.
Walking remains valuable when used to increase daily energy flux, but it functions as a complement rather than a primary driver. Low intensity movement enhances fatty acid transport and recovery without overstressing the nervous system.
Sleep and Circadian Repair Control Fat Distribution
Sleep restriction disrupts endocrine timing more severely than dietary excess. Short sleep duration increases ghrelin, decreases leptin, and elevates evening cortisol. This combination drives hunger while promoting central fat storage.
Laboratory findings summarized by the National Heart Lung and Blood Institute show sleep deficiency alters glucose metabolism similarly to prediabetes.
Melatonin secretion regulates mitochondrial repair and metabolic synchronization. Exposure to artificial light late at night suppresses melatonin and delays metabolic recovery cycles. Circadian misalignment reduces the body’s ability to oxidize fat during rest.
Deep sleep stages are when growth hormone peaks. Growth hormone stimulates lipolysis and tissue repair. Fragmented sleep eliminates this pulse, slowing fat mobilization regardless of diet or exercise quality.
Consistent sleep timing stabilizes endocrine rhythm, allowing predictable insulin sensitivity during waking hours and fat oxidation during fasting periods.
Stress Physiology and the Cortisol Abdomen
Chronic psychological stress produces continuous hypothalamic pituitary adrenal activation. Cortisol remains elevated beyond its adaptive role, directing energy storage toward the abdominal cavity.
Cortisol increases gluconeogenesis, raising blood glucose even without food intake. The pancreas responds with insulin, locking fat inside adipocytes. This cycle repeats independent of caloric consumption.
Neuroendocrine research discussed in Endotext explains how prolonged cortisol exposure changes fat distribution patterns toward visceral storage.
Sympathetic dominance also reduces digestive efficiency and thyroid signaling, lowering metabolic throughput. The body interprets chronic stress as environmental threat, prioritizing energy reserves.
Breathing patterns, physical movement, and recovery intervals influence autonomic balance. When parasympathetic activity is restored, metabolic processes resume normal regulation.
Gut Microbiome Influence on Abdominal Fat
Microbial composition affects caloric extraction, inflammation, and insulin sensitivity. Dysbiosis favors bacterial strains associated with increased energy harvest from food and elevated endotoxin production.
Endotoxins trigger low grade inflammation that interferes with insulin receptors. This state encourages fat storage despite moderate intake. Studies referenced in Nature demonstrate that microbiota composition can influence obesity independent of genetics.
Fermented foods and fiber diversity support microbial populations that produce butyrate, a short chain fatty acid that improves mitochondrial efficiency and reduces inflammation.
Antibiotic exposure, chronic stress, and highly processed diets reduce microbial diversity, weakening metabolic resilience.
Restoring microbiome balance improves nutrient partitioning so consumed energy supports tissue function rather than fat storage.
Energy Flux Versus Energy Restriction
Traditional dieting focuses on reducing intake. Metabolic restoration focuses on increasing throughput. High energy flux means the body processes more energy dynamically through movement, repair, and adaptation.
Low flux states signal scarcity, slowing thyroid activity and reducing spontaneous movement. High flux states signal abundance of activity, allowing fat stores to be used without triggering conservation mechanisms.
Athletic populations maintain low body fat percentages not because they eat minimally but because their metabolic turnover remains elevated. This principle is discussed in performance literature available through the International Journal of Sport Nutrition and Exercise Metabolism.
Increasing metabolic demand while maintaining adequate nutrition prevents adaptive slowdown commonly seen in restrictive diets.
Hormonal Interactions That Determine Fat Loss Access
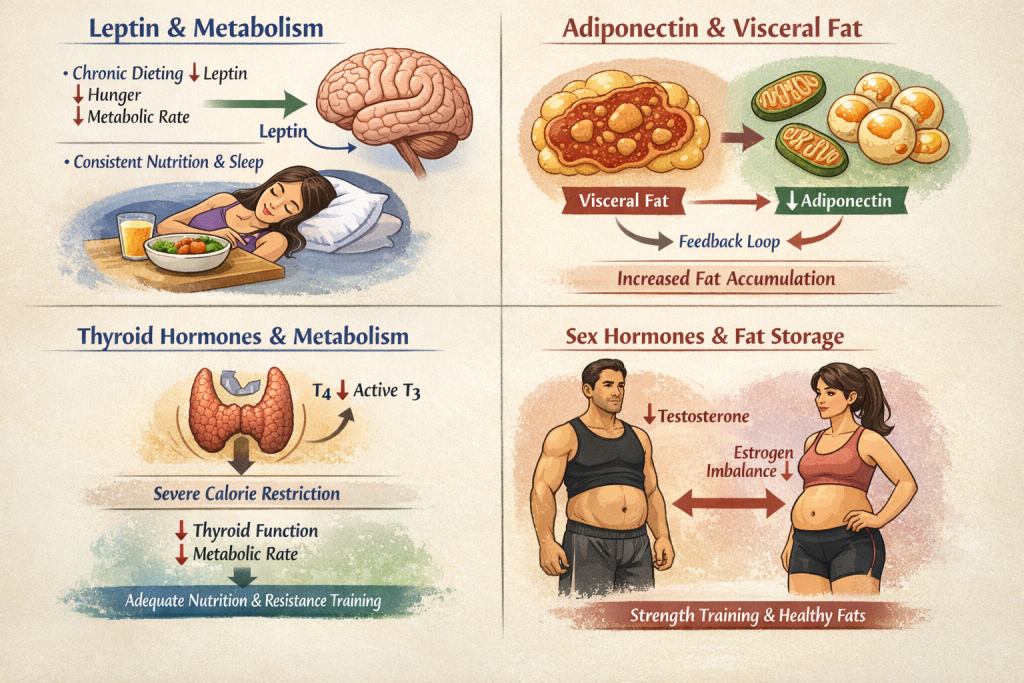
Leptin communicates energy sufficiency to the brain. Chronic dieting lowers leptin, increasing hunger and decreasing metabolic rate. Restoring leptin sensitivity requires consistent fueling patterns and sleep regulation.
Adiponectin enhances fatty acid oxidation and improves insulin sensitivity. Visceral fat suppresses adiponectin production, creating a feedback loop that favors further accumulation.
Thyroid hormones regulate basal metabolic rate. Severe calorie restriction reduces conversion of T four to active T three, slowing metabolism. Sustainable fat reduction depends on preserving thyroid signaling through adequate nutrition and resistance stimulus.
Sex hormones also influence abdominal fat storage. Reduced testosterone or estrogen imbalance shifts fat distribution centrally. Strength training and sufficient dietary fat intake support hormonal equilibrium.
Environmental Inputs That Influence Metabolic Rate
Temperature exposure affects energy expenditure. Mild cold exposure activates brown adipose tissue, which burns energy to generate heat. Brown fat activation increases total daily energy expenditure without additional exercise.
Sedentary environments suppress non exercise activity thermogenesis, the unconscious movement that contributes significantly to caloric expenditure. Standing, walking, and physical tasks restore this baseline metabolic activity.
Urbanization reduces environmental variability, contributing to metabolic rigidity. Reintroducing physical variability supports adaptive energy use.
Long Term Adaptation Versus Short Term Intervention
Short interventions fail because metabolic systems adapt only to repeated signals. Consistency reshapes enzyme expression, mitochondrial density, and hormonal rhythm.
Fat loss becomes a downstream effect of restored regulation rather than a forced outcome. When insulin sensitivity improves, muscle mass increases, sleep stabilizes, and stress decreases, abdominal fat loses its biological purpose and is mobilized naturally.
This framework explains why aggressive dieting repeatedly fails while structured lifestyle recalibration produces durable change documented across longitudinal health studies such as those tracked by the Framingham Heart Study.
Metabolic reset is therefore a systems level correction involving physiology, behavior, and environment rather than a single intervention.



